Introduction
Messenger RNA (mRNA) technology has rapidly transformed the pharmaceutical and biotechnology landscape, moving from a niche research tool to a mainstream therapeutic platform. The global success of mRNA-based COVID-19 vaccines accelerated investment, innovation, and regulatory clarity around mRNA products. At the heart of this technology lies the mRNA drug substance, often referred to as the Active Pharmaceutical Ingredient (API). Understanding what constitutes an mRNA drug substance, how it is manufactured, and why it differs from traditional small-molecule or biologic APIs is essential for professionals working in drug development, manufacturing, quality, and regulatory affairs.
Definition
mRNA Drug Substance and API refer to the purified messenger RNA (mRNA) molecule that constitutes the active pharmaceutical ingredient of an mRNA-based medicinal product. This mRNA is synthetically produced through in vitro transcription and is designed to encode a specific therapeutic protein or antigen. As the drug substance/API, it is characterized by defined sequence integrity, purity, potency, and biological activity, and it serves as the core active component prior to formulation with delivery systems such as lipid nanoparticles.
What Is an mRNA Drug Substance?
An mRNA drug substance is the purified, active nucleic acid molecule that encodes a specific protein of therapeutic or prophylactic interest. Once delivered into human cells, the mRNA is translated by the cellular machinery to produce the target protein, which then exerts the desired biological effect. In vaccines, this protein is typically an antigen that triggers an immune response, while in therapeutic applications it may replace or supplement a missing or defective protein.
Unlike conventional APIs, which are often chemically synthesized small molecules or complex recombinant proteins, mRNA is a genetic instruction set. It does not act directly on a receptor or enzyme but instead relies on the body’s own translational machinery. This unique mechanism of action places mRNA drug substances at the intersection of biologics, gene therapy, and nucleic acid–based medicines.
Key Components of an mRNA API
An mRNA drug substance is not simply a string of nucleotides. Its performance, stability, and safety depend on several carefully engineered components:
5’ Cap Structure – The 5’ cap enhances mRNA stability and ensures efficient recognition by ribosomes. Modern mRNA APIs often use synthetic cap analogs to improve translation efficiency.
5’ and 3’ Untranslated Regions (UTRs) – These regions regulate mRNA stability and translational efficiency. Optimized UTR sequences can significantly increase protein expression.
Open Reading Frame (ORF) – The ORF encodes the therapeutic protein. Codon optimization is commonly used to enhance translation without altering the protein sequence.
Poly(A) Tail – A critical element for stability and translation, the poly(A) tail length is tightly controlled during manufacturing.
Modified Nucleosides – Many mRNA APIs incorporate modified nucleosides, such as pseudouridine, to reduce innate immune activation and improve tolerability.
Together, these elements define the molecular identity and performance of the mRNA API.
Manufacturing of mRNA Drug Substance
The production of mRNA drug substance differs substantially from traditional API manufacturing. The process is primarily cell-free, relying on enzymatic reactions rather than living cell cultures.
In Vitro Transcription (IVT):
Manufacturing begins with a DNA template containing the target sequence. Using RNA polymerase, nucleotides, and specific reaction conditions, mRNA is synthesized through in vitro transcription. This step is highly scalable and allows rapid changes to the encoded sequence, one of the major advantages of mRNA technology.
Enzymatic Processing:
Following transcription, enzymatic steps are performed to add or ensure the correct 5’ cap and poly(A) tail, depending on the chosen manufacturing strategy. Some processes integrate these features directly during transcription.
Purification:
Purification is a critical step in producing a high-quality mRNA API. Impurities may include residual DNA templates, enzymes, truncated RNA species, and double-stranded RNA, which can trigger unwanted immune responses. Techniques such as chromatography and tangential flow filtration are commonly used.
Formulation as Drug Substance:
The purified mRNA, often stored in a frozen or buffered solution, constitutes the drug substance. It is later combined with a delivery system - most commonly lipid nanoparticles (LNPs) - to create the final drug product.
Critical Quality Attributes (CQAs)
As with any API, mRNA drug substances must meet strict quality standards. Key CQAs include:
Identity – Confirmation of the correct nucleotide sequence
Purity – Control of process- and product-related impurities
Integrity – Full-length mRNA without significant degradation
Potency – Ability to express the intended protein at the required level
Immunogenicity Risk – Minimization of innate immune activation
Advanced analytical methods such as capillary electrophoresis, next-generation sequencing, and in vitro translation assays are used to characterize these attributes.
Regulatory Perspective on mRNA APIs
Regulatory agencies such as the FDA and EMA classify mRNA drug substances as biological APIs. While regulatory frameworks for mRNA are still evolving, expectations are increasingly well defined. Manufacturers must demonstrate robust control over raw materials, manufacturing processes, and analytical methods.
One of the unique regulatory advantages of mRNA APIs is platform consistency. Once a manufacturing platform is established and validated, new mRNA products encoding different proteins may leverage existing data, potentially accelerating development timelines. However, each new sequence still requires thorough evaluation for safety, quality, and efficacy.
Advantages of mRNA as an API
mRNA drug substances offer several compelling advantages over traditional APIs:
Speed of Development – New mRNA sequences can be designed and manufactured rapidly
Flexibility – The same manufacturing platform can be used for multiple products
No Genomic Integration – mRNA does not integrate into host DNA, improving safety
Transient Expression – Protein production is temporary and controllable
These features make mRNA particularly attractive for vaccines, oncology, rare diseases, and personalized medicine.
Challenges in mRNA Drug Substance and API Market
Stability and Storage:
mRNA drug substances are inherently unstable and highly sensitive to temperature, enzymatic degradation, and mechanical stress. Maintaining stability throughout manufacturing, storage, and distribution often requires ultra-cold chain logistics, increasing cost and complexity.
Manufacturing Scale-Up and Consistency:
Scaling up mRNA API production while ensuring batch-to-batch consistency remains challenging. Tight control of in vitro transcription, purification, and impurity removal is critical, particularly as global demand grows.
Raw Material Availability and Quality:
High-quality nucleotides, enzymes, capping reagents, and lipids are essential for mRNA production. Limited suppliers and stringent quality requirements can create supply chain bottlenecks and increase production risks.
Regulatory and Analytical Complexity:
Regulatory frameworks for mRNA APIs are still evolving. Demonstrating robust control strategies, defining critical quality attributes, and validating advanced analytical methods require significant expertise and regulatory engagement.
Future Trends of mRNA Drug Substance and API Market
Expansion Beyond Vaccines:
mRNA drug substances are rapidly expanding beyond infectious disease vaccines into oncology, rare diseases, protein replacement therapies, and gene editing applications, significantly broadening the market potential.
Advancements in Stability and Formulation:
Innovations in sequence optimization, nucleoside modifications, and delivery systems are expected to improve mRNA stability, reduce cold-chain dependence, and enable more convenient storage and distribution.
Platform-Based Manufacturing Growth:
Standardized, platform-based manufacturing approaches will gain momentum, allowing faster development timelines, improved scalability, and cost efficiencies across multiple mRNA APIs.
Increasing Regulatory Maturity:
As regulatory agencies gain more experience with mRNA products, clearer guidelines and harmonized expectations will emerge, supporting smoother approvals and global market expansion.
Rising CDMO and Strategic Partnerships:
Growing demand for mRNA APIs will drive increased collaboration with contract development and manufacturing organizations (CDMOs), enabling companies to access specialized expertise, flexible capacity, and faster commercialization.
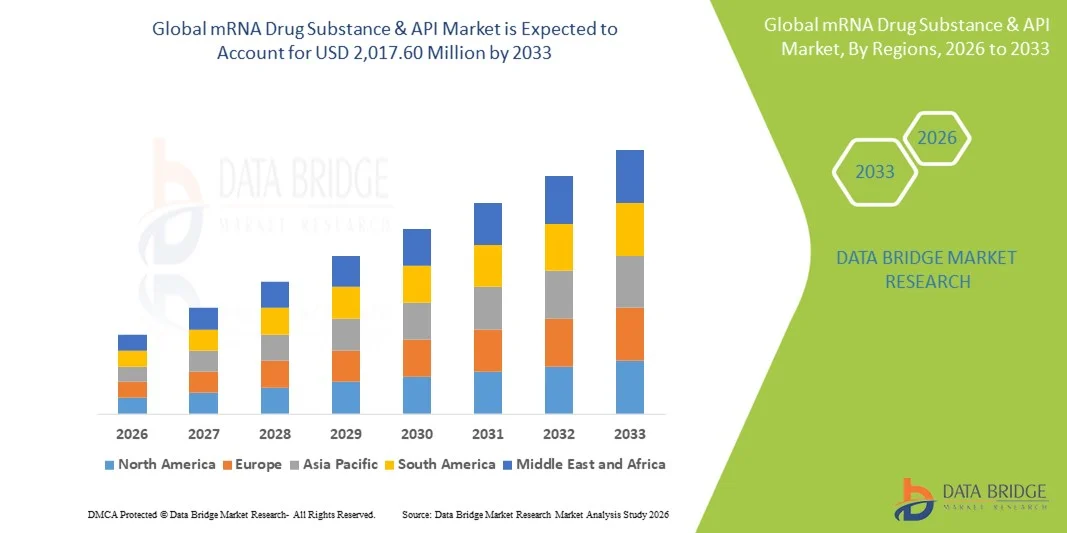
Growth Rate of mRNA Drug Substance and API Market
According to Data Bridge Market Research, the mRNA drug substance & API market was estimated to be worth USD 940.50 million in 2025 and is projected to grow at a compound annual growth rate (CAGR) of 10.00% to reach USD 2,017.60 million by 2033.
Learn More: https://www.databridgemarketresearch.com/reports/global-mrna-drug-substance-and-api-market
Conclusion
mRNA drug substance and API development represents a paradigm shift in how medicines are designed and produced. By leveraging the body’s own protein synthesis machinery, mRNA APIs offer unparalleled speed, flexibility, and therapeutic potential. While challenges related to stability, manufacturing, and regulation remain, ongoing innovation continues to address these issues. For pharmaceutical companies, regulators, and healthcare providers alike, understanding mRNA drug substances is essential as this transformative technology reshapes the future of drug development.
Powered by Froala Editor
You may also like
More from this category.
What Is the Fastest Way to Cure Nail Fungus?
Finding the Right Vein Treatment Doctor Hamden Patients Can Trust

Why Visiting a Spider Vein Specialist Astoria Patients Trust Can Improve Leg Comfort and Confidence

How Infertility Treatment Can Improve Your Chances of Conception

Hospital Furniture Supplier in India – ALIS PRO

Innovative Regenerative Care for Healthier Hair Growth Exosome Hair Therapy in Islamabad:
Does Ozempic Injection speed up strong body transformation wave?

Blood Tests in Delhi | Home Sample Collection
Pharmacy-led digestive care is shaping everyday self-treatment in France

